- #ISO 13485 AUDIT CHECKLIST HOW TO#
- #ISO 13485 AUDIT CHECKLIST UPDATE#
- #ISO 13485 AUDIT CHECKLIST ISO#
During the first phase, auditors will review your quality management system, evaluate site-specific conditions, and activate discussions with key stakeholders. The objective of the Stage 1 Audit is to determine an organization's readiness for Stage 2 Certification Audit. Perform Stage 1 and 2 Certification Auditįinally, you'll need to perform appropriate certification audits.

#ISO 13485 AUDIT CHECKLIST UPDATE#
You may need to revisit documented procedures to determine whether or not a software update is required. It's the process and supporting documentation to ensure that your software meets regulatory requirements and guidelines.ĭuring the validation phase, it's important to develop appropriate change controls to monitor the validated state.
#ISO 13485 AUDIT CHECKLIST ISO#
Validation is a critical first-step toward eQMS software adoption, implementation, and ISO 13485:2016 certification. RELATED: The 7 Best Online Quality Management Training Courses 4. Organizations also need to quantify and measure an employee's ability to perform in their role.Īs such, organizations are required to create a list of skill requirements for each position, establish criteria for successful training, and produce evidence that employees have met minimum requirements for training, testing, and certification. In fact, it's no longer enough to document that employees have completed their training requirements. However, ISO 13485:2016 makes it clear that training activities should be more rigorous than simply checking a box. ISO 13485:2016 includes a subtle expansion of training requirements.Įffective training is an important component of any quality management system. As such, the less complicated the process, the easier it will be to ensure that all stakeholders can produce routine quality outcomes. These documents ensure that quality systems are understood and repeatable across the entire ecosystem. Keep in mind that the quality management system documentation consists of both the mandatory documents and other documents specified by regulatory bodies. By doing so, you can identify any significant gaps that might be preventing your organization from aligning with ISO 13485 requirements. Next, define the quality processes and procedures that your life science organization currently abides by. RELATED: 5 Tips to Get ISO 13485 / ISO 14971 Certification 2. As a best practice, consider investing in an electronic quality management systems (eQMS) that delivers the following features: This enables you to know the exact requirements necessary to maintain compliance. Start by familiarizing yourself with ISO 13485 regulations. ISO 13485:2016 is the most up-to-date standard built-in response to the latest quality management processes.įollow this checklist to receive ISO 13485 certification and comply with the latest standards.
We are now prepared to take next steps towards compliance with European regulatory requirements," said Jonas T., Quality Manager.Įvery five years, ISO standards are reviewed to determine whether a revision is required in order to keep it current within the marketplace.
"Without Qualio, we would have not been able to achieve our critical milestone of ISO 13485 certification before the end of 2015.

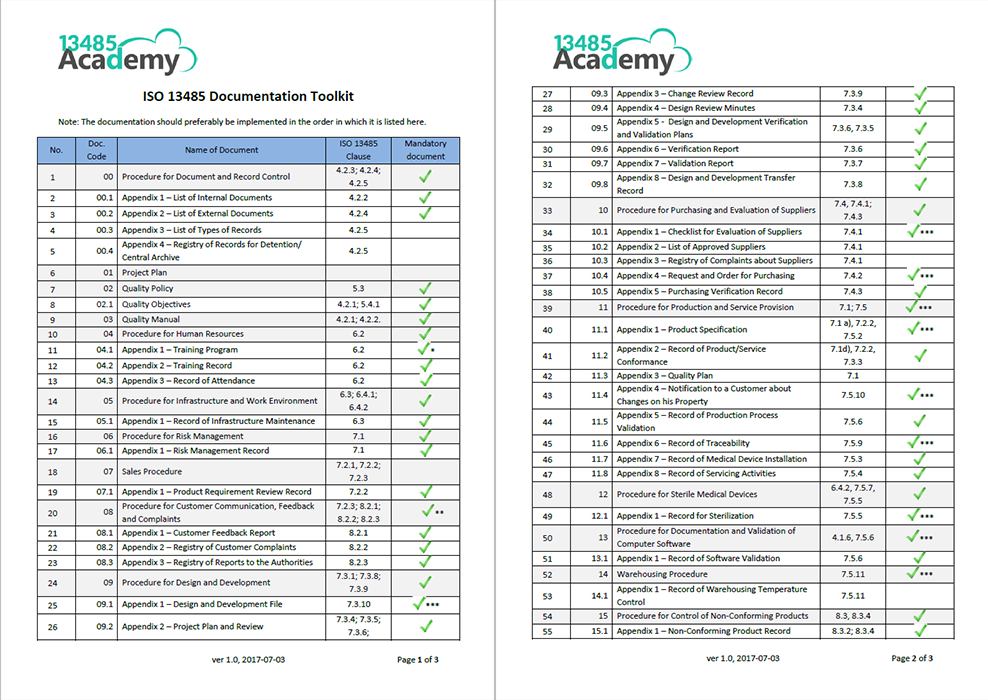
The ISO 13485 Checklist for Medical Device ManufacturersĪs an industry-leading quality management system partner, we've helped hundreds of life science organizations orient towards rapid ISO 13485 success. Having a proven and systematized process helps streamline the entire certification process.įor this reason, we've created a simple ISO 13485 checklist for medical device manufacturers.Ĭonsult this helpful guide as you prepare for ISO 13485 certification. Without proper certification preparation and documentation, you could end up wasting valuable time, energy, and capital. To comply, organizations need to demonstrate their ability to produce medical devices that meet customer and regulatory requirements. ISO 13485 outlines the unique requirements for a quality management system (QMS).
#ISO 13485 AUDIT CHECKLIST HOW TO#
Keep reading to learn more about how to make the experience easier for your medical device company. Are you preparing for ISO 13485 certification? If so, the process can be overwhelming.
